23. The nuclide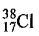 decays by B-emisson with a half-life of 37.2 min. A sample of0.80 mol of H38Cl is placed in a 11.0 L closed container at 300 K, where one of the decay products is H2(g). What is the pressure (atm) in the container after 74.4 min? (R=0.082 atm-L.mol-¹.K-¹)
decays by B-emisson with a half-life of 37.2 min. A sample of0.80 mol of H38Cl is placed in a 11.0 L closed container at 300 K, where one of the decay products is H2(g). What is the pressure (atm) in the container after 74.4 min? (R=0.082 atm-L.mol-¹.K-¹)
(A) 1.23
(B) 1.79
(C) 2.24
(D) 2.46
(E) 3.13
答案:登入後查看
統計: A(1), B(2), C(0), D(0), E(0) #3847799
統計: A(1), B(2), C(0), D(0), E(0) #3847799