41. Using the following data reactions: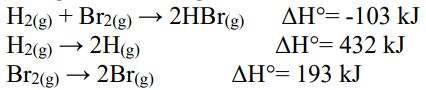
calculate the energy of an H-Br bond.
(A) 728 kJ
(B) 261 kJ
(C) 364 kJ
(D) 522 kJ
(E) 182 kJ
答案:登入後查看
統計: A(0), B(1), C(5), D(2), E(0) #3840051
統計: A(0), B(1), C(5), D(2), E(0) #3840051