60. The Henderson-Hasselbalch equation (below) is widely used in biochemistry to calculate the pH of buffer solutions. When 100 mL of a 0.1 mM buffer solution made from acetic acid (pKa is 4.76.) and sodium acetate with pH 5.0 is diluted to 1 liter, what is the pH of the diluted solution? 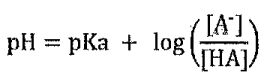
(A) 4.50
(B) 4.76
(C) 5.00
(D) 5.32
(E) 5.54
答案:登入後查看
統計: A(0), B(1), C(2), D(0), E(1) #3847776
統計: A(0), B(1), C(2), D(0), E(1) #3847776