62. Baking powder, a mixture of cream of tartar (KHC4H4O6, molar mass 188 g/mol) and baking soda (NaHCO3, molar mass 84.0 g/mol), undergoes the following reaction at baking temperature: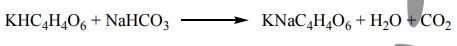
(The CO2 makes the cake rise.) A recipe calls for two level teaspoons (a total of 8.0g) of cream of tartar. How much baking soda must be added for both materials to react completely?
(A) 0.45 g
(B) 1.8g
(C) 3.6g
(D) 8.0 g
(E) none of these
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料