1. The reaction: CH3CHCH2(g) + HCl(g) →CH3CHClCH3(g) and the possible mechanism is

Complex + (HCl)2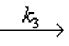 CH3CHClCH3 + 2HCl Slow equilibrium
CH3CHClCH3 + 2HCl Slow equilibrium
Derive the rate law for this reaction using this mechanism.
1. The reaction: CH3CHCH2(g) + HCl(g) →CH3CHClCH3(g) and the possible mechanism is

Complex + (HCl)2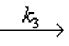 CH3CHClCH3 + 2HCl Slow equilibrium
CH3CHClCH3 + 2HCl Slow equilibrium
Derive the rate law for this reaction using this mechanism.