1. Compare the two reaction coordinate diagrams below and select the answer that correctly describes their relationship. In eachcase, the single intermediate is the ES complex.Free energy,
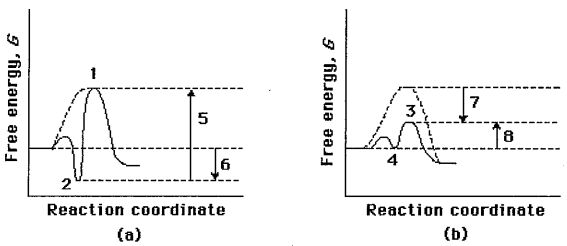
(A) Diagram (a) describes a strict "lock and key" model, whereas diagram (b) describes a transition-state complementarity model.
(B) The activation energy for the catalyzed reaction is #5 in diagram (a) and is #7 in diagram (b).
(C) The activation energy for the uncatalyzed reaction is given by #5 + #6 in diagram (a) and by #7 + #4 in diagram (b).
(D) The contribution of binding energy is given by #5 in diagram (a) and by #7 in diagram (b).
(E) The ES complex is given by #2 in diagram (a) and #3 in diagram (b).
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料