13. The Electromotive Force (emf) ofthe cell Pt,H2(lbar)|HCI(0.01m) AgCl(s)|Ag is 0.2002V at 25°C, 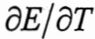 is –8.665×10-5V/K.
is –8.665×10-5V/K.
[Hint: The electrode reactions of cell are 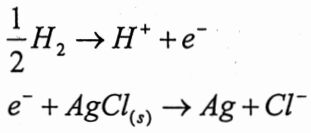 ;The cell reaction is
;The cell reaction is 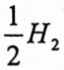 + AgCl(s) → Ag + H+ +Cl-; e =1.602x10-19C]
+ AgCl(s) → Ag + H+ +Cl-; e =1.602x10-19C]
The Gibbs energy change of the cell reaction is △ G =
(A) -1.932 J/mol ,
(B) -19.32 J / тol ,
(C) -193.2 J/mol,
(D) -1,932 J/mol ,
(E) -19,320 J/ mol
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料