34. When a student performs an endothermic reaction in a calorimeter, how (if any) does the calculated value of AH differ from the actual value if the heat exchanged with the calorimeter is not taken into account?
(A) 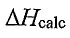 is less positive because the reaction absorbs heat from the calorimeter.
is less positive because the reaction absorbs heat from the calorimeter.
(B) 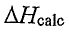 is less negative because the calorimeter absorbs heat from the reaction.
is less negative because the calorimeter absorbs heat from the reaction.
(C)  is more positive because the reaction absorbs heat from the calorimeter.
is more positive because the reaction absorbs heat from the calorimeter.
(D)  is more negative because the calorimeter always absorbs heat from the reaction.
is more negative because the calorimeter always absorbs heat from the reaction.
(E) 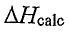 equals the actual value because the calorimeter does not absorb heat. :
equals the actual value because the calorimeter does not absorb heat. :
答案:登入後查看
統計: A(1), B(0), C(0), D(0), E(0) #3477639
統計: A(1), B(0), C(0), D(0), E(0) #3477639