(b) Calculate the density in g/cm3 of UO2 (uranium oxide), which has the calcium fluoride
structure. (Ionic radii: 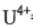 = 0.11 nm and
= 0.11 nm and 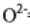 = 0.13 nm; the atomic mass of uranium is 238
amu and oxygen is 16 amu).
= 0.13 nm; the atomic mass of uranium is 238
amu and oxygen is 16 amu).
(b) Calculate the density in g/cm3 of UO2 (uranium oxide), which has the calcium fluoride
structure. (Ionic radii: 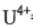 = 0.11 nm and
= 0.11 nm and 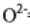 = 0.13 nm; the atomic mass of uranium is 238
amu and oxygen is 16 amu).
= 0.13 nm; the atomic mass of uranium is 238
amu and oxygen is 16 amu).