(5-2)∫∫∫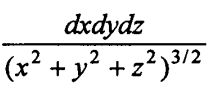 , 其中 b2 ≦ (x² + y² + z²) ≦ a²
, 其中 b2 ≦ (x² + y² + z²) ≦ a²
24. Considering parallel reactions forming P1 and P2 from A, choose the correct statement given
the potential energy diagram on the right side.
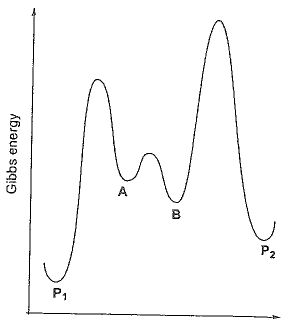 (A) There is an intermediate from A to P2.
(B) P2 is the kinetically controlled product.
(C) With a catalyst significantly increasing the rate from B
to P2, the selectivity towards P2 can be improved by
lowering reaction temperature.
(D) There are three transition states in this potential energy
diagram.
(E) P2 is the thermodynamically controlled product.
(A) There is an intermediate from A to P2.
(B) P2 is the kinetically controlled product.
(C) With a catalyst significantly increasing the rate from B
to P2, the selectivity towards P2 can be improved by
lowering reaction temperature.
(D) There are three transition states in this potential energy
diagram.
(E) P2 is the thermodynamically controlled product.
23. Given a sequential reaction below, choose the correct statement assuming k1 <k2.
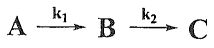 (A) At the same conversion, the selectivity of B can be improved by increasing the reaction
temperature.
(B) The |m| (m = slope) of the curve of the first reaction is smaller than that of the second
reaction in the Arrhenius plot.
(C) With the same reaction time, adding a catalyst to catalyze the second reaction can improve
the yield of C.
(D) The apparent activation energy of the whole sequential reaction is closer to the activation
energy of the second reaction.
(E) The second reaction has a lower activation energy.
(A) At the same conversion, the selectivity of B can be improved by increasing the reaction
temperature.
(B) The |m| (m = slope) of the curve of the first reaction is smaller than that of the second
reaction in the Arrhenius plot.
(C) With the same reaction time, adding a catalyst to catalyze the second reaction can improve
the yield of C.
(D) The apparent activation energy of the whole sequential reaction is closer to the activation
energy of the second reaction.
(E) The second reaction has a lower activation energy.
22. He(g) (n mole) is taken from state 1 (P1, V1) to state 4 (P4, V4) through state 2 (P2, V2) or
state 3 (P3, V3). Assuming He(g) obeys ideal gas law, choose the correct statement.
(A) w = 0 from state 2 to state 4.
V.-30 L
(B) q= 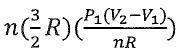 from state 1 to state 2. (C)△H = n
from state 1 to state 2. (C)△H = n 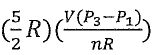 from state 1 to state 3. (D) △H = q from state 3 to state 4.
(E) Pathway A (through state 2) and pathway B (through state 3) have the same △H.
from state 1 to state 3. (D) △H = q from state 3 to state 4.
(E) Pathway A (through state 2) and pathway B (through state 3) have the same △H.