10. Consider the following Lewis structure. (Lone pairs are not drawn in.)
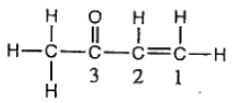
Which statement about the molecule is incorrect?
(A)There are 10 \(\sigma\) and 2 \(\pi\) bonds.
(B)Oxygen is sp3 hybridized.
(C)C-2 is sp2 hybridized with bond angles of 120°.
(D)This molecule contains 28 valence electrons.
(E)There are some H–C–H bond angles of about 109° in the molecule.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料