30. The rate law for the following mechanism is
Cl2(g) ⇌ 2 Cl(g) both fast, equilibrium constant K1,
Cl(g) + CO(g) ⇌ COCl(g) both fast, equilibrium constant K2
COCl(g) + Cl2(g) → COCl2(g) + Cl(g) k3, slow
(A) 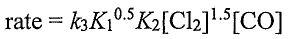
(B)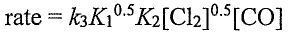
(C) rate = k3K1K2Cl2
(D) rate = k3CO
(E) rate = K1K2k3[CO]
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料