47. The photochemical chlorination of chloroform, CHCl3 + Cl2 → CCl4 + HCl is believed to proceed by the following mechanism:
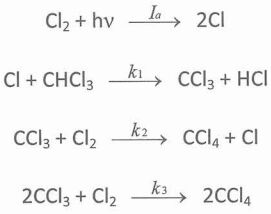
Ia represents the intensity of light absorbed: number of photons absorbed per unit volume per unit second. The rate law for the production of carbon tetrachloride is d[CCl4]/dt =
(A) k2Ia1/2[Cl2]1/2/k31/2 + 2Ia
(B) k1Ia1/2[Cl2]1/2/k21/2 + 2Ia
(C) k3Ia1/2[Cl2]1/2/k21/2 + Ia
(D) 2k2Ia1/2[Cl2]1/2/k31/2 + 2Ia
(E) k2Ia[Cl2]3/2/k31/2 + Ia
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料