13. If 2.019 g of ethanol C2H5OH(I) is burned completely in a bomb calorimeter at 298.15 K, the heat produced is 59.68 kJ. △Hf0 for water(1) =-285.8 kJ mol-1 and for CO2(g) =-393.5 kJ mol-1.
(A) 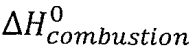 for ethanol(l) at 298.15 K is 886.7 kJ mol-1
for ethanol(l) at 298.15 K is 886.7 kJ mol-1
(B) 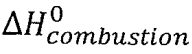 for ethanol(1) at 298.15 K is –886. kJ mol-1
for ethanol(1) at 298.15 K is –886. kJ mol-1
(C)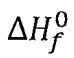 for ethanol(1) at 298.15 K is 210.5 kJ mol-1
for ethanol(1) at 298.15 K is 210.5 kJ mol-1
(D)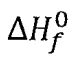 for ethanol(1) at 298.15 K is -210.5 kJ mol-1
for ethanol(1) at 298.15 K is -210.5 kJ mol-1
(E) 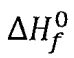 for ethanol(1) at 298.15 K is –280.4 kJ mol-1
for ethanol(1) at 298.15 K is –280.4 kJ mol-1
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料