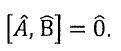3. In quantum mechanics, the measurements of two physical quantities, a and b, are represented by operators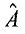 and
and 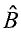 , respectively. If the two operators commute, which of the following statements is NOT true? (A) It is possible to measure a and b precisely and simultaneously. (B) The two operators possess a common set of eigenfunctions. (C) The two operators possess a common set of eigenvalues. (D)
, respectively. If the two operators commute, which of the following statements is NOT true? (A) It is possible to measure a and b precisely and simultaneously. (B) The two operators possess a common set of eigenfunctions. (C) The two operators possess a common set of eigenvalues. (D)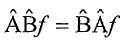 , where f is a well-behaved function. (E)
, where f is a well-behaved function. (E)