24. A reaction is catalyzed by the bromide ion shown below: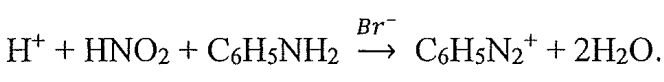
A proposed mechanism is
(1) H++ HNO2 ⇌ H2NO2+
(2) H2NO2++Br-→ ONBr + H2O
(3) ONBr + C6H5NH2 → C6H5N2+ + H2O + Br-
Symbol kn denotes the rate constant for the step n.
(A) Assume that the second step is the rate-limiting. The rate law is k2[H2NO2+][Br_]
(B) Assume that the second step is the rate-limiting. The rate law is k2[H+][HNO2][Br-]
(C) Assuming quasi-steady-state approximation, the rate law is k2[H2NO2+][Br-].
(D) Assuming quasi-steady-state approximation and excess [Br- ], the rate law is k1[H+][HNO2]
(E) Assuming quasi-steady-state approximation and excess [Br-], the rate law is (k1/k-1) k2[H+][HNO2][Br-]
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料