7. Consider the variational method for the ground state of a two-electron atom. The simplest trial function is the Hartree product of the form 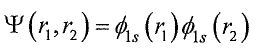 ), where
), where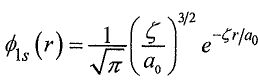 isthe hydrogen-like satomic orbitalwith anefective nuclear changes, ζ , as the variational parameter. The variational integral can be evaluated analytically to give
isthe hydrogen-like satomic orbitalwith anefective nuclear changes, ζ , as the variational parameter. The variational integral can be evaluated analytically to give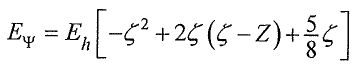 , where Z is the true nuclear charge and Eh=27.2 eV is the Hartree energy.What is the ground state energy of Li+ ion at this level of approximation?
, where Z is the true nuclear charge and Eh=27.2 eV is the Hartree energy.What is the ground state energy of Li+ ion at this level of approximation?
(A) -77.5 eV
(B) -196.5 eV
(C) -54.4 eV
(D) -108.8 eV
(E) -98.2 eV
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料