9. The complex form of H-atom wave functions for n=3, 1=2, m=±1 states are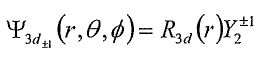 ,where R3d(r) is the radial function and
,where R3d(r) is the radial function and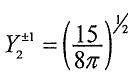 (sinθcosθ)e±iφ are spherical harmonic functions with 1=2 and m=±1. A real form function (3dxz orbital) can be constructed with the linear combination of the two complex wave functions as
(sinθcosθ)e±iφ are spherical harmonic functions with 1=2 and m=±1. A real form function (3dxz orbital) can be constructed with the linear combination of the two complex wave functions as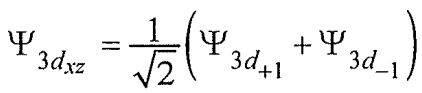 . Which of the following statements about the 3dxz orbital is NOT correct?
. Which of the following statements about the 3dxz orbital is NOT correct?
(A) 3dxz wave function is an eigenfunction ofH-atom 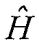 (Hamiltonian operator).
(Hamiltonian operator).
(B)3dxz wave function is an eigenfunction of H-atom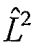 (operator for square of orbital angular momentum).
(operator for square of orbital angular momentum).
(C) 3dxz wave function is an eigenfunction of H-atom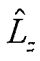 (operator for angular momentum along the z axis).
(operator for angular momentum along the z axis).
(D) The magnitude of orbital angular momentum is not zero.
(E) the average values of Lz is zero.
統計: 尚無統計資料