25. At constant temperature, consider a equilibrium reaction,
3 Fe(s) + 4 H2O(g) ⇌ 4 H2(g) + Fe3O4(s). If the volume of the container is reduced
(A) 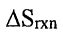 is increased.
is increased.
(B) 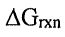 is decreased.
is decreased.
(C) 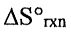 is increased.
is increased.
(D) 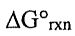 is decreased.
is decreased.
(E) The above answers are all incorrect.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料