46. The wavefunction of one of the 3d orbitals for the hydrogen atom is 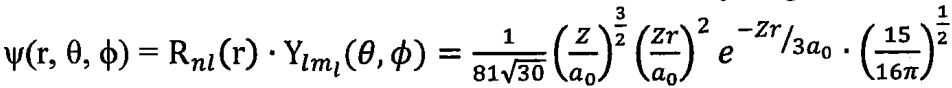 sin²θsin2φ where Rnl(r) is the radial function and Ylᵐl(θ,φ) is the angular function. How many radial nodes and angular nodes are there in this orbital?
sin²θsin2φ where Rnl(r) is the radial function and Ylᵐl(θ,φ) is the angular function. How many radial nodes and angular nodes are there in this orbital?
(A) 0 and 1
(B) 0 and 2
(C) 1 and 1
(D) 1 and 2
(E) 2 and 2
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料