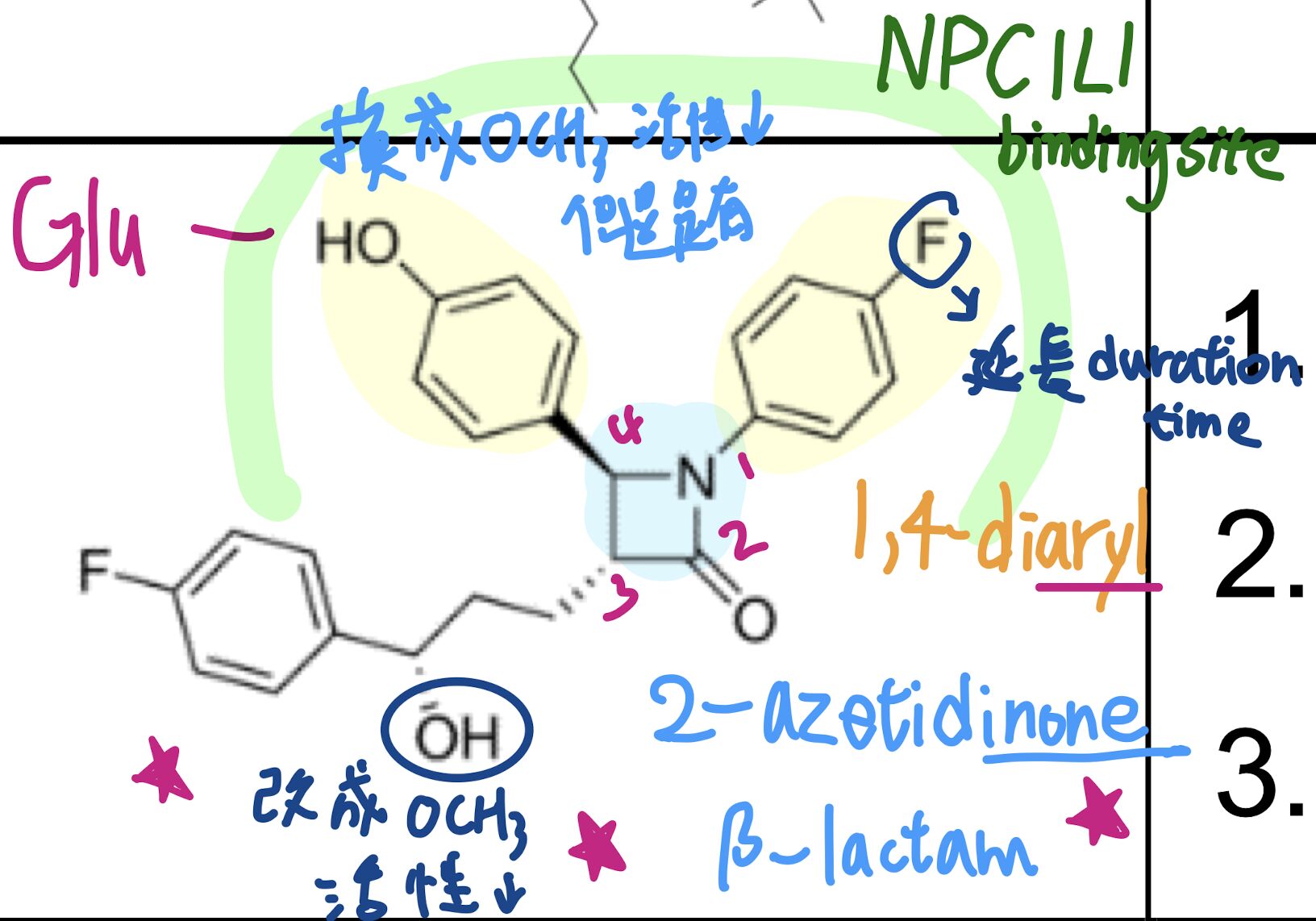
47.下列有關對 ezetimibe 結構與活性之敘述,何者錯誤?
(A)具有β-lactam 結構
(B)phenolic 基團改成 p-methoxyphenyl 仍有活性
(C)aliphatic hydroxyl 改成 aliphatic methoxy 則活性提高
(D)p-fluorophenyl 之 fluoro 取代,可延長作用時間
統計: A(605), B(1211), C(2626), D(709), E(0) #2866785
詳解 (共 8 筆)
提供大家自己的一點想法~~(有錯再請大家糾正> <
因為Ezetimibe 具glucuronide的活性代謝物
所以需要OH 才能binding
所以OH是一定要留的喔!
抱歉我只有7版QQ
原文:
Using compound D as a lead, as well as in vivo data suggesting that metabolic transformations produced the active compound ultimately responsible for the cholesterol-lowering effect, structural modificationswere made that, eventually, led to the development of ezetimibe. The most important changes involved the introduction of hydroxyl groups to help localize the compound in
the intestine and the introduction ofp-fluoro groups to block undesirable metabolism. (P831)
大概就是 Compound D 是雛形,然後改良成 ezetimibe,然後 OH 可以讓 ezetimibe 在腸道更專一、p-fluoro可以延長作用時間,然後因為 Compound D 本來就有 p-Methoxy且有活性,所以 (B) phenolic 基團改成 p-methoxyphenyl 仍有活性是對的、(C) aliphatic hydroxyl 改成 aliphatic methoxy 則活性提高 --> 降低
然後前面有提到還有一個 Compound C,原本是設計抑制 ACAT (acyl CoA–cholesterol acyltransferase) 的
結論:
p-Methoxy、苯環接 OH:Improve
苯環接 Methoxy:仍有活性

小碎念:我連查課本都查半天了,到底誰會 = =
Ezetimibe 考古
46.下列何者為ezetimibe的結構?