10. Three possible Lewis structures (①, 2 and ③) can be drawn for nitrous oxide (N2O). Which of the following statements regarding N₂O and its Lewis structures is correct? 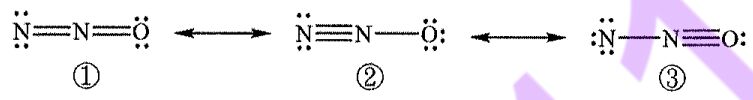
(A) The oxidation state of nitrogen in N2O is +2.
(B) The formal charge on the oxygen atom is the same in all three Lewis structures.
(C) Based on formal-charge considerations, ①is the most fayorable.
(D) Based on formal-charge considerations, ② is the most favorable.
(E) The terminal nitrogen atom in ③ is sp-hybridized.
答案:登入後查看
統計: A(0), B(0), C(4), D(0), E(0) #3847786
統計: A(0), B(0), C(4), D(0), E(0) #3847786