11. Pt(NH3)2Cl₂ has two isomers,① and② , as shown in the figure below. Compound① is called cisplatin, and compound ②is called transplatin. One of them is an anticancer drug, and one is inactive. The anticancer drug works by its chloride ions undergoing a substitution Cl NH3 CI NH reaction with nitrogen atoms in DNA Pt Pt that are close together, forming a N-Pt-N angle of about 90°. Which of the following statements regarding the structures and properties of isomers ①and ② is correct? 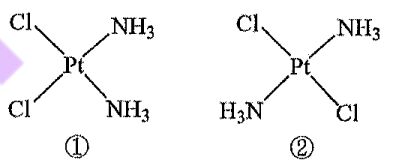
(A) The types of chemical bonds present in Pt(NH3)2C2 are coordinate (dative) bonds and ionic bonds.
(B) The d-orbital splitting patterns of the metal center in① and② are identical.
(C) Compound ② is the anticancer drug.
(D)① and② have identical polarity.
(E) IR spectroscopy can be used to distinguish between ① and②.
統計: A(0), B(1), C(1), D(0), E(2) #3847787