42. The figure represents a closed cycle for an ideal gas (the figure is not drawn to scale). The change in the internal energy of the gas as it moves from a to c along the path abc is -500 J. As the gas moves from state c to state d, 400 J of heat is transferred to the system. An additional 200 J of heat is absorbed by the gas as it moves from d back to a. What is the work done on the gas as it moves from c to d? 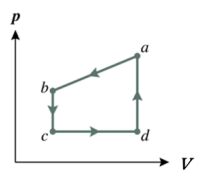
(A) -100 J
(B) 100 J
(C) -300 J
(D) 300 J
(E) -700 J
答案:登入後查看
統計: A(1), B(0), C(0), D(0), E(0) #3853557
統計: A(1), B(0), C(0), D(0), E(0) #3853557