22. Consider a hydrogen-like atom, assuming the electron orbits the nucleus of effective charge 3.7e in a simple planar circular motion, as described by the Bohr model. If the radius of the circular motion is r=2.2r0 where r0=0.529×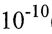 (m). What is the magnetic moment of this hydrogen-like atom? [1/4π∈0 =8.99×109(N m²/Coul²)]
(m). What is the magnetic moment of this hydrogen-like atom? [1/4π∈0 =8.99×109(N m²/Coul²)]
(A)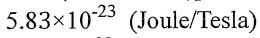
(B)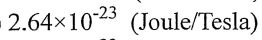
(C)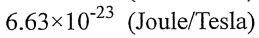
(D)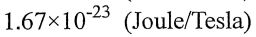
答案:登入後查看
統計: A(0), B(1), C(0), D(0), E(0) #3829857
統計: A(0), B(1), C(0), D(0), E(0) #3829857