25. An ideal monatomic gas undergoes an adiabatic process that the pressure and volume change from (P1, V1) to (P2, V2). What is the work done in this adiabatic process?
(A)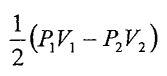
(B)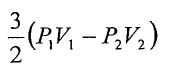
(C)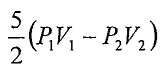
(D)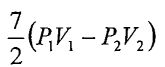
(E)None of the above
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料