3. One mole of ideal monatomic gas is taken around the reversible cycle as shown in the following figure. The isothermals are at 500 K and 300 K. (R is the gas constant)
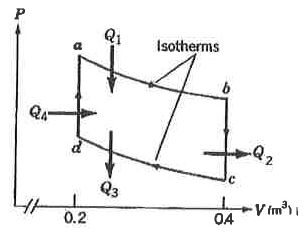
(A) Q₁ = 500 Rln2
(B) Q₂ = −300 R
(C) Q₃ = −300 Rln2
(D) the work done in one cycle is 300 Rln2
(E) the efficiency of the engine is 0.214.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料