6. Consider a thermodynamic engine based on ideal gas. The operating cycle is shown in the pressure(P)-volume(V) diagram. The a→b and c→d are isochoric (constant volume) processes, and the b→c and d→a are adiabatic processes. Which one of the following statements is wrong?
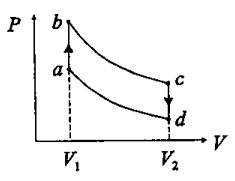
(A) During a→b, the heat flows from the engine to the surrounding.
(B) During b→c, the temperature of the gas decreases.
(C) During c→d, the internal energy of the gas decreases.
(D) During d→a, the internal energy of the gas increases.
(E) During d→a, the engine does negative work on the surrounding.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料