7. The relation PV = nRT holds for all ideal gases. The additional relation \( PV^\gamma \) holds for an adiabatic process. The figure below shows two curves: one is an adiabat and one is an isotherm. Each starts at the same pressure and volume. Which statement is correct? (Note: "∝" means "is proportional to".)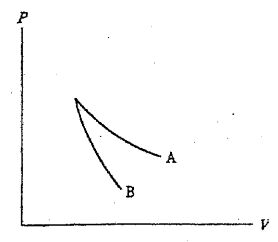
(A)Isotherm: \( P \propto \frac{1}{V} \); Adiabat: \( P \propto \frac{1}{V^\gamma} \): A is both an isotherm and an adiabat.
(B)Isotherm: \( P \propto \frac{1}{V^\gamma} \); Adiabat: \( P \propto \frac{1}{V} \): B is an isotherm, A is an adiabat.
(C)Isotherm: \( P \propto \frac{1}{V} \); Adiabat: \( P \propto \frac{1}{V^\gamma} \): A is an isotherm, B is an adiabat.
(D)Isotherm: \( P \propto \frac{1}{V^\gamma} \); Adiabat: \( P \propto \frac{1}{V} \): B is both an isotherm and an adiabat.
(E)I cannot answer this without additional information about the starting temperature.
統計: 尚無統計資料