題組內容
2. Figure 1 shows a reversible cycle through which 0.2 mol of a monatomic ideal gas is taken. Volume Vd = 4.0Va, Vb = Va, and Vc = Vd. Process bc is an isothermal expansion, with pb = 10.0 atm (=1.013×10⁶ Pa) and Vb = 1.0 × 10⁻³ m³. Process da is adiabatic (pVᵞ = constant with γ = Cp/CV) with pa = 5.0 atm. For the cycle, find (a) the temperature at point a, Ta: (3)______, (b) the energy added to the gas as heat, (4)______ (c) the efficiency of the cycle, (5)______, and (d) the entropy change ΔS during process bc, (6)______.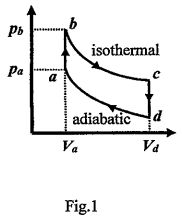
3.