2. Right figure shows a reversible cycle through which 1 mol of a monatomic ideal gas is taken. Assume p = 2p₀, V = 2V₀, p₀ = 1.01 × 10⁵ Pa, and V₀ = 0.0225 m³.
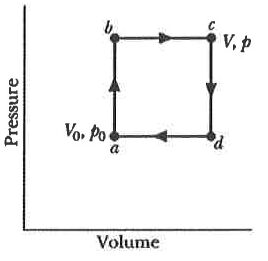
(A) The work done during the cycle is 2.27 kJ.
(B) The energy added as heat during stroke abc is 14.8 kJ.
(C) The efficiency of the cycle is 0.217.
(D) The efficiency of a Carnot engine operating between the highest and lowest temperatures that occur in the cycle is 0.25.
(E) The efficiency of Carnot engine is thus greater than the efficiency of the cycle showed in the right figure.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料