20. The figure shows the energy levels for an electron in a finite potential energy well. If an electron in the n = 2 state absorbs a photon of wavelength 2.0 nm, what happens to the electron?
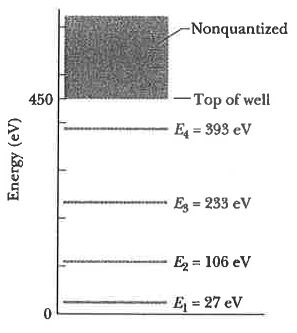
(A) It makes a transition to the n = 3 state.
(B) It makes a transition to the n = 4 state.
(C) It escapes the well with a kinetic energy of 280 eV.
(D) It escapes the well with a kinetic energy of 730 eV.
(E) Nothing; this photon does not have an energy corresponding to an allowed transition so it is not absorbed.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料