30. A boron-carbon-nitrogen compound with a diamond cubic-like structure can be synthesized under high-temperature and high-pressure conditions. Its crystal structure was determined by X-ray crystallography and its unit-cell structure is shown in the figure on the right. The cubic unit-cell parameter is a pm, and NA denotes Avogadro's constant. Which of the following statements is incorrect? (molar atomic mass (g/mol): B = 11, C = 12, N =14)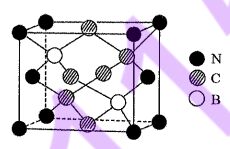
(A) This material is a covalent compound with high hardness.
(B) The number of boron atoms that are nearest neighbors to a given boron atom in the crystal and are at equal distances is 4.
(C) The ratio of the number of C-C bonds to C-N bonds in the unit cell is 2:1.
(D) The density of the unit cell is 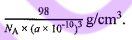
(E)The empirical formula of this compound is BC2N.
統計: A(1), B(1), C(1), D(1), E(0) #3847806