26. For an atom with one valence electron in a p-subshell (l = 1, s = 1/2), which statement about spin-orbit coupling is correct?
(A) Spin-orbit coupling splits the p-level into j = 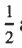 and j =
and j = 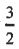 states.
states.
(B) The only allowed total angular momentum is j = 1/2 because s = 1/2.
(C) Spin-orbit coupling does not occur unless the atom is placed in an external magnetic field.
(D) The degeneracy of the j = 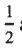 level is 4 and the degeneracy of the j =
level is 4 and the degeneracy of the j =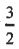 level is 2.
level is 2.
(E) Spin-orbit coupling combines n and l into a new quantum number j while leaving s unchanged.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料