29. -1. The A bipolar-membrane electrolyzer is used to reduce KNO3 to produce NH3, as shown in the figure below. The Faradaic efficiency for ammonia, FE(NH3), can electron required to form P reach 90% [Faradaic efficiency: 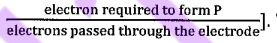 .TheFaradaic efficiency for producing other gas(es) is 50%. During operation, H2О dissociates in the interfacial (middle) layer of the bipolar membrane to generate H+ and OH-, which migrate toward the two electrodes under the electric field. Which of the following statements is correct?
.TheFaradaic efficiency for producing other gas(es) is 50%. During operation, H2О dissociates in the interfacial (middle) layer of the bipolar membrane to generate H+ and OH-, which migrate toward the two electrodes under the electric field. Which of the following statements is correct?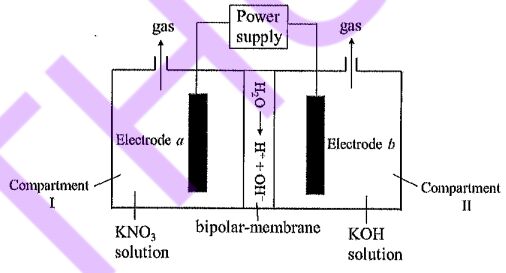
(A) Electrode potential: the potential of electrode a is higher than that of electrode b.
(B) The OH produced by water dissociation in the bipolar membrane migrates toward electrode a.
(C) The overall reaction in compartment I is: NO3+8 e+ 6 H2O → NH3 +9 OH.
(D) When 11.2 L of gas is produced at the anode (at standard conditions), 0.45 mol of NH3 is produced at the cathode.
(E) Hydrogen and oxygen gases are evolved from compartment II.
統計: A(0), B(0), C(2), D(1), E(0) #3847805