21. At room temperature, 0.10 M NaOH solution is added dropwise to 20 mL of 0.10 M CH3COOH solution. The relationship between the negative logarithm of the H+ concentration originating from the autoionization of 5- water, -log[H+]water, and the volume of 10 20 30 40 VNOH(ag/nL NaOH solution added is shown in the graph on the right. Assuming that the volume change upon mixing can be neglected, which of the following statements is correct?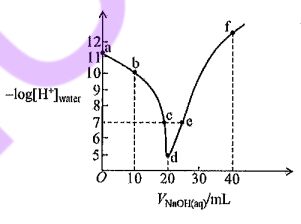
(A) At point b, [CH3COO-] < [CH3COOH].
(B) At points c and e, the solutions are both neutral.
(C) At point d, the concentration of CH3COO- is greater than that at point e.
(D) At point d, [CH3COO-] + [CH3COOH] = 0.05 M.
(E) At point f, [Na+] > [CH3COO-]<[OH-]>[ H+].
答案:登入後查看
統計: A(1), B(1), C(0), D(1), E(1) #3847797
統計: A(1), B(1), C(0), D(1), E(1) #3847797