26. The rate law for the reaction 2 H2(g) + 2 NO(g) →2 H2O(g) +N2(g) is rate = H2]a[NO]β, and the rates of the reaction under different conditions are as follow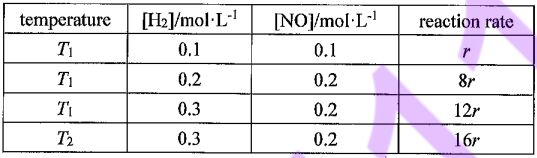
Which of the following statements is correct?
(A) T2 <T1
(B) α = 2, β = 1
(C) When all other conditions are held constant and only the concentrations of the four substances in the reaction system are varied, only the change in the concentration of the reactants has an effect on rate, and the effect of [NO] is greater.
(D) At T2, [NO] = 0.1 mol·L-1, the reaction rate is 8r, then [H2] =0.3 mol·L-1
(E) According to the varied reaction rates above, this is an endothermic reaction.
答案:登入後查看
統計: A(0), B(0), C(2), D(0), E(2) #3847802
統計: A(0), B(0), C(2), D(0), E(2) #3847802